FDA approves Eversense E3 6-month continuous glucose monitor that requires fewer fingerstick blood glucose measurements - NotebookCheck.net News
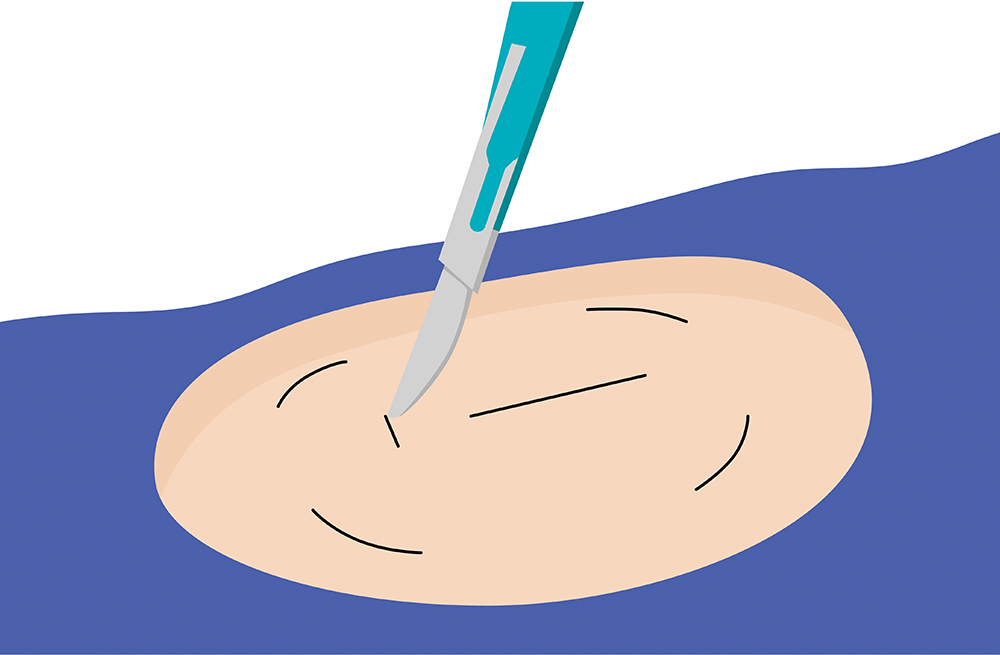
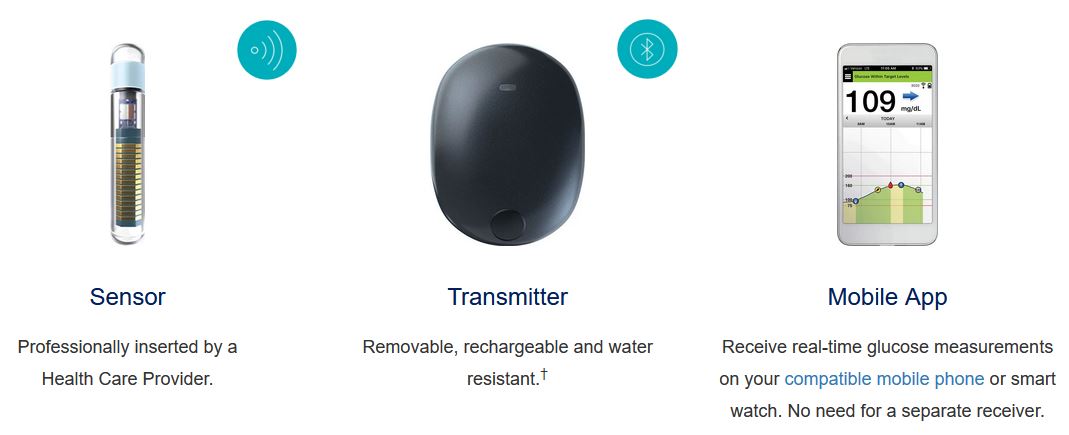
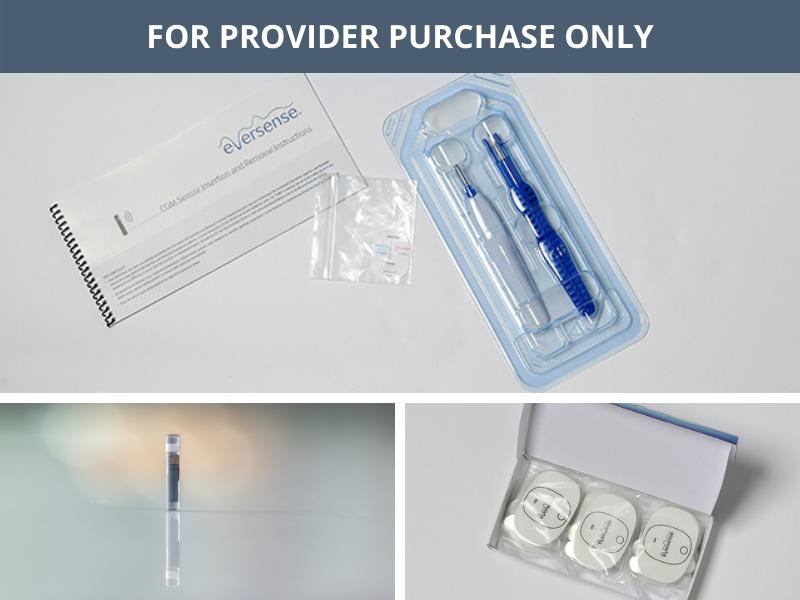
Break free from frequent and sometimes painful self-insertions weekly or bi-weekly. The Eversense sensor is carefully placed under the skin by a trained health care provider and lasts up to 6 months.

Eversense E3 CGM Approved for Two Sensors per Year: Your “Happily Ever(sense) After” - Taking Control Of Your Diabetes®

ASCENSIA DIABETES CARE LAUNCHES THE 6 MONTH EVERSENSE® E3 CONTINUOUS GLUCOSE MONITORING SYSTEM IN THE U.S. WITH THE NEW EVERSENSE PASS SAVINGS PROGRAM



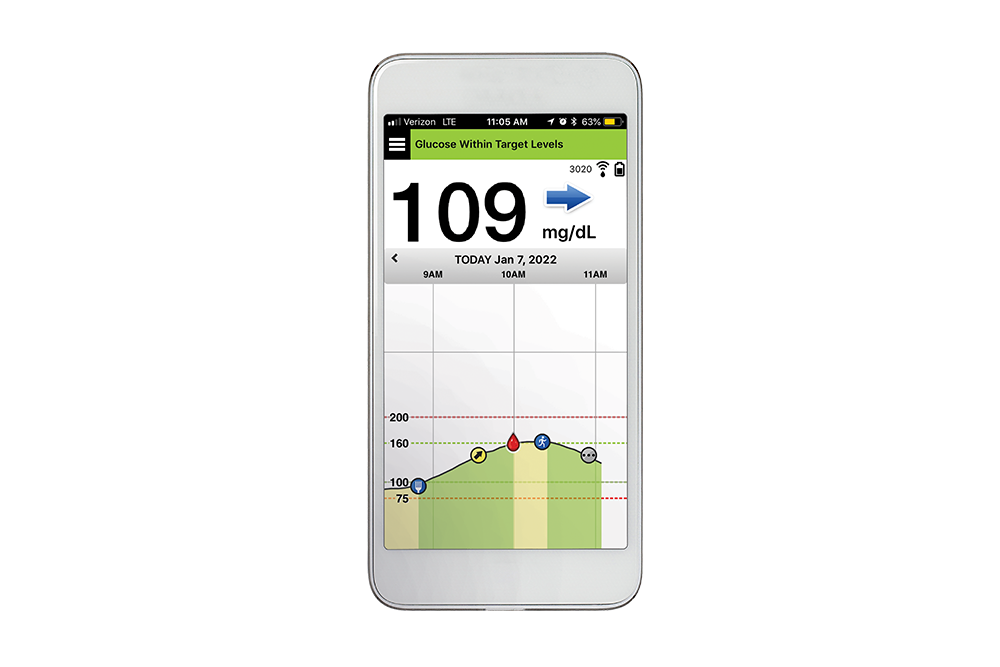





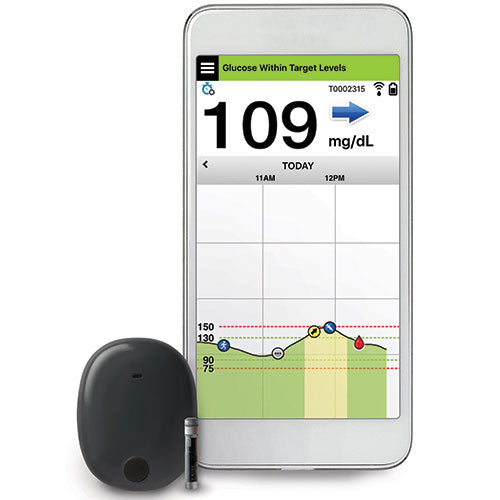







![6-Month Glucose Implant Sensor Receives CE Mark Approval [video]|Health Tech Insider 6-Month Glucose Implant Sensor Receives CE Mark Approval [video]|Health Tech Insider](https://i0.wp.com/healthtechinsider.com/wp-content/uploads/Eversense_CGM_with_video_600x275.jpg?fit=600%2C276&ssl=1)
.jpg)