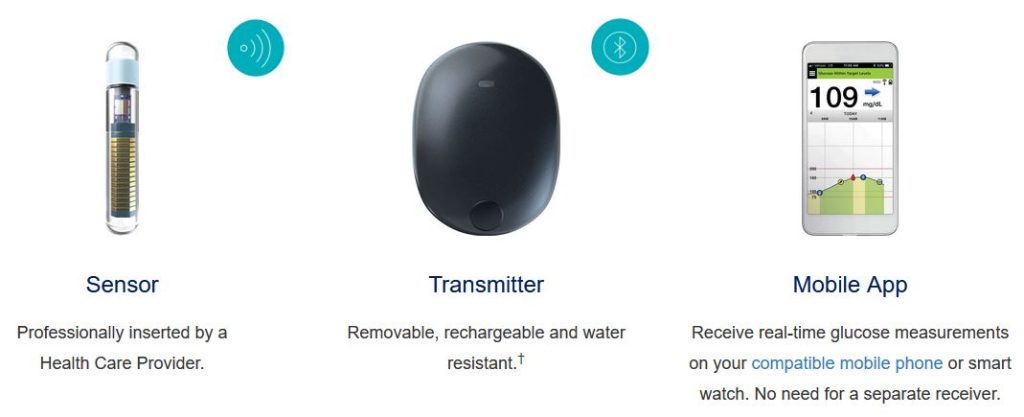
FDA approves Eversense E3 6-month continuous glucose monitor that requires fewer fingerstick blood glucose measurements - NotebookCheck.net News

Germantown's Senseonics Announces FDA Approval of the World's First and Only Long-Term Implantable Glucose Monitoring System - The MoCo Show
/assets/images/provider/photos/2654529.jpg)



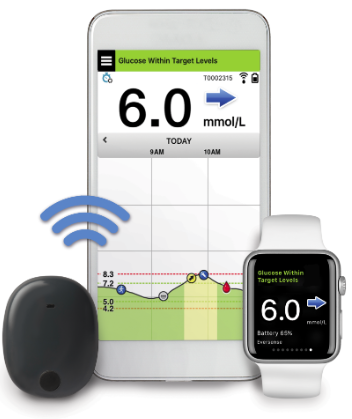
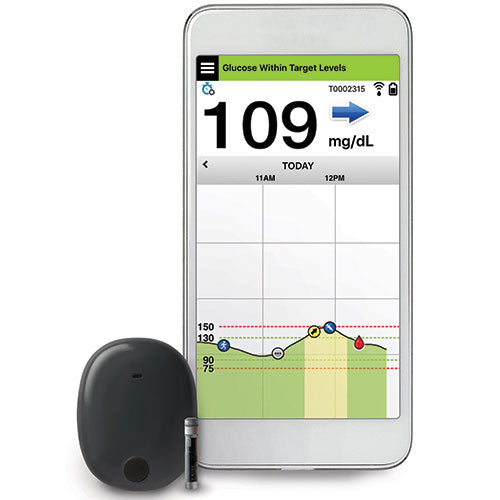





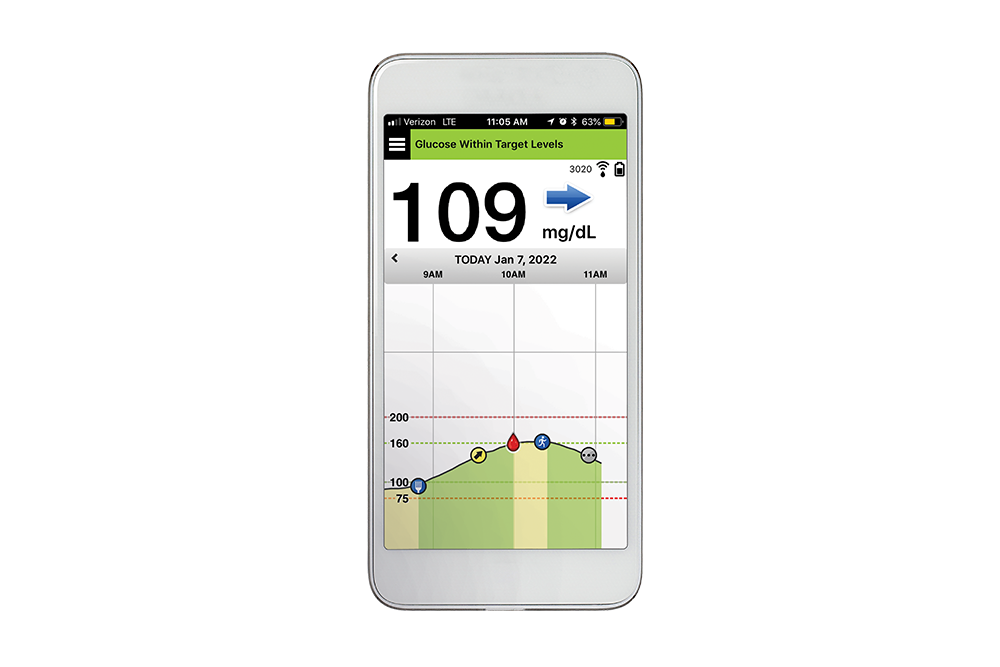






![6-Month Glucose Implant Sensor Receives CE Mark Approval [video]|Health Tech Insider 6-Month Glucose Implant Sensor Receives CE Mark Approval [video]|Health Tech Insider](https://i0.wp.com/healthtechinsider.com/wp-content/uploads/Eversense_CGM_with_video_600x275.jpg?fit=600%2C276&ssl=1)